Fats are a type of lipid, which is broadly defined as a molecule that is insoluble in water but that dissolves in organic solvents like ether and chloroform. Lipids encompass fats and oils, fatty acids, phospholipids, and sterols like cholesterol.
Lipids perform three primary biological functions within the body:
- they serve as structural components of cell membranes,
- they are an energy storage molecule and some can be directly used for energy,
- they function as important signaling molecules, including forming the backbone of autocrine (within a cell) and paracrine (between neighboring cells) signaling molecules as well steroid hormones and vitamin D.
The impact of dietary fat on our health is dependent on both the type of fat and how much of it we’re eating.
The Building Blocks: Fatty Acids
Fats and oils are more technically called triglycerides (or triacylglycerol), which are composed of three fatty acids linked together (esterified) by a glycerol molecule. Fatty acids are the building blocks of fats. They are used not only for energy but also for many basic structures in the human body, such as the outer membrane of every single cell. There are many different fatty acids, each with different effects on, and roles in, human health.
Each fatty acid consists of a bunch of hydrocarbons (a carbon atom bonded with one to three hydrogen atoms, the number of which varies for different fats) bonded together in a string, called the hydrocarbon chain, with a carboxyl group at one end, the molecular formula of which is COOH (one carbon atom bound to two oxygen atoms and a hydrogen atom).
Fatty acids can be categorized based on the type of molecular bond between the carbons in the hydrocarbon chain (saturated, monounsaturated and polyunsaturated) and by the length of the hydrocarbon chain (short-chain, medium-chain and long-chain).
Saturated, Monounsaturated and Polyunsaturated Fatty Acids
This terminology relates to the type of molecular bonds between carbon atoms in the hydrocarbon chain part of the fatty acid molecule.
Saturated Fats
A saturated fatty acid is one in which all the bonds between carbon atoms in the entire hydrocarbon chain are single bonds (a simple molecular bond in which two adjacent atoms share a single electron). The carbons are then also “saturated” with hydrogen atoms, meaning that each carbon atom in the middle of the chain is bound to two hydrogen atoms. What’s special about saturated fatty acids is that they are very stable and not easily oxidized (which means they are not prone to react chemically with oxygen).
Monounsaturated Fats
A monounsaturated fatty acid is one in which one of the bonds between two carbon atoms in the hydrocarbon chain is a double bond (that is, a molecular bond in which two adjacent atoms share two electrons). This double bond replaces two hydrogen atoms, so the hydrocarbon chain is no longer “saturated” with hydrogen. Monounsaturated fats are less stable than saturated fats and require more enzymes to break apart in order to be used as energy than saturated fats do.
Polyunsaturated Fats
A polyunsaturated fatty acid is one in which two or more of the bonds between carbon atoms in the hydrocarbon chain are double bonds (again, replacing hydrogen atoms in the chain). Polyunsaturated fats are easily oxidized, meaning that they are prone to react chemically with oxygen. This reaction typically breaks the fatty acid apart and produces oxidants (free radicals).
Short-, Medium- and Long-Chain Fatty Acids
This terminology refers to the length of the hydrocarbon chain part of the fatty acid molecule.
- Short-chain fatty acids are those with fewer than 6 carbon atoms in the hydrocarbon chain, notable examples of which include acetic acid (2-carbon), propionic acid (3-carbon), and butyric acid (4-carbon). Short-chain fatty acids do not esterify into triglycerides. They are found in fermented foods and some dairy products.
- Medium-chain fatty acids are those with 6 to 12 carbon atoms in the hydrocarbon chain, and include caproic acid (6-carbon), caprylic acid (8-carbon), capric acid (10-carbon), and lauric acid (12-carbon). If you’ve heard the terms medium chain triglycerides, this refers to three medium-chain fatty acids esterified together into a triglyceride molecule. They are found in palm and coconut products.
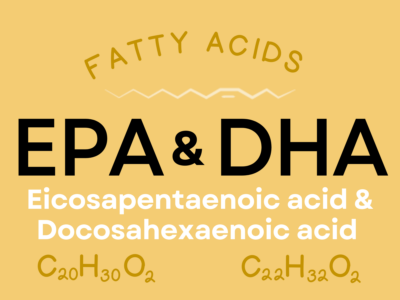
- Long-chain fatty acids are those with more than 12 carbon atoms in the hydrocarbon chain, some notable examples of which are linoleic acid (18-carbons), alpha-linolenic acid (18-carbon), oleic acid (18-carbon), eicosapentaenoic acid (20-carbon) and docosahexaenoic acid (22-carbon). Most of the fats found in foods are long-chain.
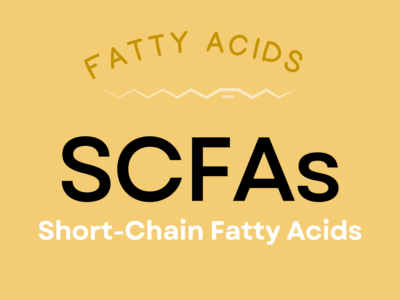
Short-Chain Fatty Acids (SCFAs)
Short-chain fatty acids (SCFAs) are unique fats produced by the bacterial fermentation of carbohydrates in the gut, with smaller amounts coming directly from diet. They serve as the main energy source for cells lining the colon, giving them an important role in gastrointestinal health—including a potentially protective effect against colorectal cancer. They also influence appetite and metabolism, mineral absorption, the function of the immune system, and insulin and blood glucose regulation! Through an impact on the gut-brain axis, SCFAs can even affect mood and neurological health.
Medium-Chain Triglycerides (MCTs)
Medium-chain triglycerides (MCTs) are a type of saturated fat composed of at least two medium-chain fatty acids. Their exceptionally rapid and direct absorption (straight from the intestine to the liver) allows them to be quickly burned for fuel. The earliest use of MCTs was to help treat epilepsy, but they also possess benefits for weight loss and body composition—including by spontaneously reducing appetite and food intake, and by increasing resting energy expenditure via thermogenesis (heat production). MCTs may also help increase insulin sensitivity among diabetics, improve memory and cognition in Alzheimer’s patients, improve exercise performance, and boost gut health.
Omega-3 and Omega-6 Fatty Acids Are Essential
Polyunsaturated fats are also broadly categorized as omega-3 fatty acids and omega-6 fatty acids. These classifications relate to the location of the first double bond in relation to the end of the hydrocarbon tail. If the first double bond is between the third and fourth carbon atoms, it’s an omega-3 fatty acid. If it’s between the sixth and seventh, it’s an omega-6 fatty acid.
Our bodies can convert any omega-6 polyunsaturated fatty acid to any other omega-6 polyunsaturated fatty acid, and similarly can convert any omega-3 polyunsaturated fatty acid to any other omega-3 polyunsaturated fatty acid. So, the shortest-chain omega-3 and omeag-6 fatty acids and the ones assigned as the essential fatty acids: linolenic acid (ALA; the shortest omega-3 polyunsaturated fatty acid) and linoleic acid (LA; the shortest omega-6 polyunsaturated fatty acid). However, the term essential is misleading here because conversion from one omega-6 to another, or from one omega-3 to another, can be extremely inefficient. The fatty acids with the most profound roles in the human body are arachidonic acid (AA), an omega-6 polyunsaturated fatty acid, and eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), both omega-3 polyunsaturated fatty acids, so it’s important to get these from foods. While ALA and LA are abundant in plant foods, AA, EPA, and DHA are found in seafood, meat, and poultry.
Cis and Trans Fats
This terminology refers to the orientation of the hydrogen atoms on either side of a double bon between carbon atoms in the hydrocarbon chain part of the fatty acid molecule.
The double bond between carbons in naturally-occurring monounsaturated and polyunsaturated fats is most commonly in the cis configuration, where the two hydrogen atoms on either side of the bond are on the same side, putting a kink in the fatty acid. Many man-made fats and a few notable naturally-occurring trans fats (like conjugated linoleic acid) have double bonds between carbon atoms in the trans configuration, where hydrogen atoms are on opposite sides, creating a straighter molecule.
Commercially, trans fats are formed by taking a polyunsaturated oil (like soybean oil) and injecting it with hydrogen to make it more solid (a process called hydrogenation). This converts some of the cis double bonds to trans double bonds, resulting in partially hydrogenated oils that are high in trans fats. Because of their texture and long shelf life (saturating some of the double bonds makes these fats less susceptible to oxidation), hydrogenated oils were used extensively by food manufacturers to make cheap, shelf-stable products. The highest levels of trans fats are found in margarines, processed snack foods, frozen dinners, commercial baked goods, and fast food.
But, what’s good for the food industry isn’t necessarily good for human health! In numerous studies, trans fats have been linked to higher LDL “bad” cholesterol, lower HDL “good” cholesterol, and higher inflammation in a dose-dependent effect (that is, the more trans fat we eat, the more harmful the changes in these risk factors). In mice, trans fats have been shown to reduce the responsiveness of a growth factor called TGF-β1 that helps control the growth, proliferation, differentiation, and apoptosis of cells. Population studies consistently link higher consumption of trans fats directly to heart disease. And, some research shows a link between trans fats and diabetes, likely due to the effect on insulin sensitivity and glucose transport when these fats are integrated into cell membranes. Several studies have even suggested that trans fats could raise the risk of certain cancers!
Conjugated Linoleic Acids (CLA)
Conjugated linoleic acid (CLA) is an omega-6 fatty acid mostly found in meat and dairy products from grass-fed animals. Despite technically being a trans fat, CLA exhibits a range of beneficial health properties, including anti-cancer, anti-obesity, anti-diabetes, and anti-heart disease activities. It also promotes gut health and immune function.
Everything You Need to Jump into Nutrivore TODAY!

Nutrivore Quickstart Guide
The Nutrivore Quickstart Guide e-book explains why and how to eat a Nutrivore diet, introduces the Nutrivore Score, gives a comprehensive tour of the full range of essential and important nutrients!
Plus, you’ll find the Top 100 Nutrivore Score Foods, analysis of food groups, practical tips to increase the nutrient density of your diet, and look-up tables for the Nutrivore Score of over 700 foods.
Buy now for instant digital access.
How Much Dietary Fat Do We Need?
Excessive fat intake is associated with increased risk of obesity, diabetes, cardiovascular disease, dementia, certain cancers and even certain autoimmune diseases. Of course, it’s not as simple as “go low-fat,” which comes with its own health risks like obesity, diabetes, cardiovascular disease, depression and anxiety, certain cancers, depressions, and certain autoimmune diseases. (Yes, those lists are nearly identical! Mind blown, right?) It turns out that a huge variety of factors are at play here, like the type of fat, the overall quality of the diet, whether the diet includes excess calories, and whether the person is overweight.
The Food and Nutrition Board of the Institute of Medicine has set Accepted Macronutrient Distribution Ranges (AMDR) for protein, fat, and carbohydrates based on evidence from interventional trials with support of epidemiological evidence that suggests a role in the prevention or increased risk of chronic diseases and based on ensuring sufficient intake of essential nutrients. The AMDR for fat is estimated to be 20% to 35% of total energy. This Goldilocks range (not low fat, not high-fat) is likely optimal both for our gut microbiomes and our metabolic health.
No RDA is established for total fat or individual fatty acids; however, the World Health Organization recommends an omega-6 fatty acid intake of 2.5–9% of total calories and an omega-3 fatty acid intake of 0.5–2% of calories .
Nutrivore Is a Game-Changer—This FREE Guide Shows You Why
Sign up for the free Nutrivore Newsletter, your weekly, science-backed guide to improving health through nutrient-rich foods — without dieting harder —and get the Beginner’s Guide to Nutrivore delivered straight to your inbox!

Citations
Expand to see all scientific references for this article.
Hu FB, et al. “Diet, lifestyle, and the risk of type 2 diabetes mellitus in women.” N Engl J Med. 2001 Sep 13;345(11):790-7.
Ibrahim A, et al. “Dietary trans-fatty acids alter adipocyte plasma membrane fatty acid composition and insulin sensitivity in rats.” Metabolism. 2005 Feb;54(2):240-6.
Mozaffarian D, et al. “Health effects of trans-fatty acids: experimental and observational evidence.” Eur J Clin Nutr. 2009 May;63 Suppl 2:S5-21.
Natarajan S, et al. “Dietary trans fatty acids alter diaphragm phospholipid fatty acid composition, triacylglycerol content and glucose transport in rats.” Br J Nutr. 2005 Jun;93(6):829-33.
Help me to keep building Nutrivore.com

I’m working on writing more awesome articles on this topic to be featured right here on this webpage. If you’re enjoying the content my team and I have created thus far and would like to see more, you can support our efforts to keep building out this site by joining the Nutrivore community on Patreon!
Plus every month, you’ll gain exclusive and early access to a variety of resources, including a weekly video podcast, a new e-book in a series, nutrient fun factsheet, and more! Sign up now and also get 5 free Nutrivore guides as a welcome gift! Win-win-win!